There is no intention here to imply that any of the procedures discussed here are safe, legal or even sane. I take no responsibility for what anyone may do with this information. The &%$^#@ environmentalists have made the process of refrigeration into a big Three Ring Circus. The regulations that they have brought about are so stringent that one should never underestimate the ease of being a criminal when involved with any refrigeration process. Yes, There are high pressures involved, and I know that propane (R 290) is flammable and explosive and that there are many reasons that a system of this nature may not always be suitable for domestic uses. So Please spare me from letters and comments about my stupidity. This is not a plan for a practical, safe, reliable refrigerator. It's sole purpose was to observe a working refrigeration process using materials obtained from the nearest hardware store. The materials used are crude by refrigeration standards and the possibility of leaks are high, making this a project to strongly consider being done out of doors.
There are some fairly common substances that make good refrigerants. Propane is one of them and is classified as R 290. The ban on R 12 greatly intensifies the fascination of being able to use ordinary propane, from the hardware store, as a refrigerant in a home built experiment. I decided to build a crude system of my own and see if I could get something frosty in my own garage. Sources say that propane is compatible with the mineral oil that is used in R-12 systems. Any information on this subjuct is certainly welcome. I felt that the project would be a total success if It could cause even a tiny spot, anywhere on the system, to get frosty. My best expectations were greatly exceeded when the entire cold side of the system, including all the fittings and valves, frosted up in just two or three minutes. It makes the kind of cold that grabs your fingers when touched.
Before starting with the system described below, I did a simple experiment using just a can of propane, and a thermometer. The cannister of propane was fitted with a valve from a used brazing kit and had a rubber hose. The thermometer was of the glass mercury type that red down to -30 deg F. I pointed the rubber hose at the bulb of the thermometer and opened the valve for a few seconds, spraying it with liquid propane. The thermometer fell, in a few seconds, below the -30 deg f mark and went clear down into the bulb.
The next step was to make the refrigeration system after observing the dramatic refrigerant quality that propane has. There was one exception made to using common hardware store materials and that was in making use of an existing refrigerator pump that I had laying around for more than a decade. I went to the hardware store and purchased 15 feet of 1/4" copper tubing, 4 small brass valves and various fittings etc. I also got a 14 oz can of propane, the kind used on a blowtorch for soldering copper pipe.
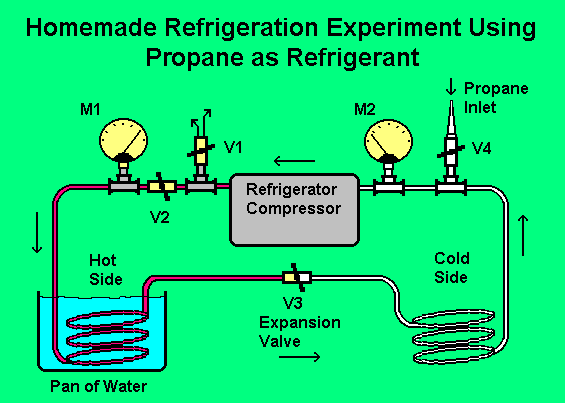
The picture below shows how it was built. The 15 foot length of copper tubing was chosen arbitrarily and cut in the middle; a valve (V3) was inserted to be used as the expansion valve. This is the valve that takes the place of the normally used capillary tube. The valve has to be adjusted all the time as the temperature changes and is quite critical. This is one of many reasons why this system is not about to replace the refrigerator in my kitchen. The adjustment is easy though because of the two meters installed. The reason for having the other three valves is to make the system capable of evacuating itself prior to the addition of the propane. Since I didn't have a special cooling radiator, the coiled tubing on the hot side was immersed in a pan of tap temperature water to keep it cool. The picture is mostly self explanatory and a list of operation procedures is given below.

The tubing on the right is the same as that on the left, but looks white because it is covered with frost.

1. Start by evacuating the system. V1 and V3 open, V2 and V4 closed. Run the pump for a few minutes. At first air will be exiting from V1. After a while it will diminish to nothing. The meter that I used for M2 happened to be the type that also reads vacuum. The system evacuated to below 20".
2. Close V1 and open V2. The system is now running with most of the air removed. Connect the propane cannister through a high pressure rubber hose to the inlet barb on V4. Having a fitting on the cannister with a valve, as described above, is helpful. Open V4 and the valve on the propane cannister until both meters read about 70 or 80 psig. V4 and the valve on the propane cannister can now be closed and the rubber hose removed from the barb on V4. The system is now charged.
3. Close V3 most of the way until you get a reading on M2 of between 15 and 20 psig. This seems to be the optimal settings on my system. M1 should rise to 200 or more psig. Within seconds, the copper tube on the low pressure side of V3 should start to frost up. As the system runs, you will need to keep on top of the pressure setting on the meters by adjusting V3. The system has been a thrill to operate in spite of this. Maybe later, I will find an improved form of V3; it is a very crude expansion control. After a while, the water that is cooling the hot side will get warm and the efficiency of the system will diminish, making a change of water desirable.
I weighed the propane cannister before and after filling the system. Using a kitchen food scale, I could see that approx 1 oz. of propane had been used.
The psig pressure readings given for the meter readings above, mean pressure in pounds per square inch as compared to atmospheric pressure. This is called gauge (g) scale. Pressure readings sometimes are given as psia. This means pressure in pounds per square inch as compared to an absolute (a) vacuum. Thus, the difference between a reading listed as psig and one listed as psia would be roughly 14 to 15 psi. The psia reading would be higher because it includes atmospheric pressure.
Propane is compressed by the compressor into the hot side or high pressure side tubing. The propane, like any gas tends to get hot. If the tubing is kept cool, the high pressure propane will condense under the pressure into a liquid. Condensing tends to make it even hotter, but the idea is to cool it down; in this case we are using water. This tubing full of room temperature liquid is now the same thing as the cannister full of propane. When the expansion valve is opened just a little, the high pressure liquid propane squirts into the cold or low pressure side tubing and vaporizes; just like it did when squirting it on to the thermometer. Any time a liquid vaporizes, it tends to absorb heat and make things, that it comes in contact with, very cold. The vapor finally finds its way to the input side of the pump where it is compressed again.